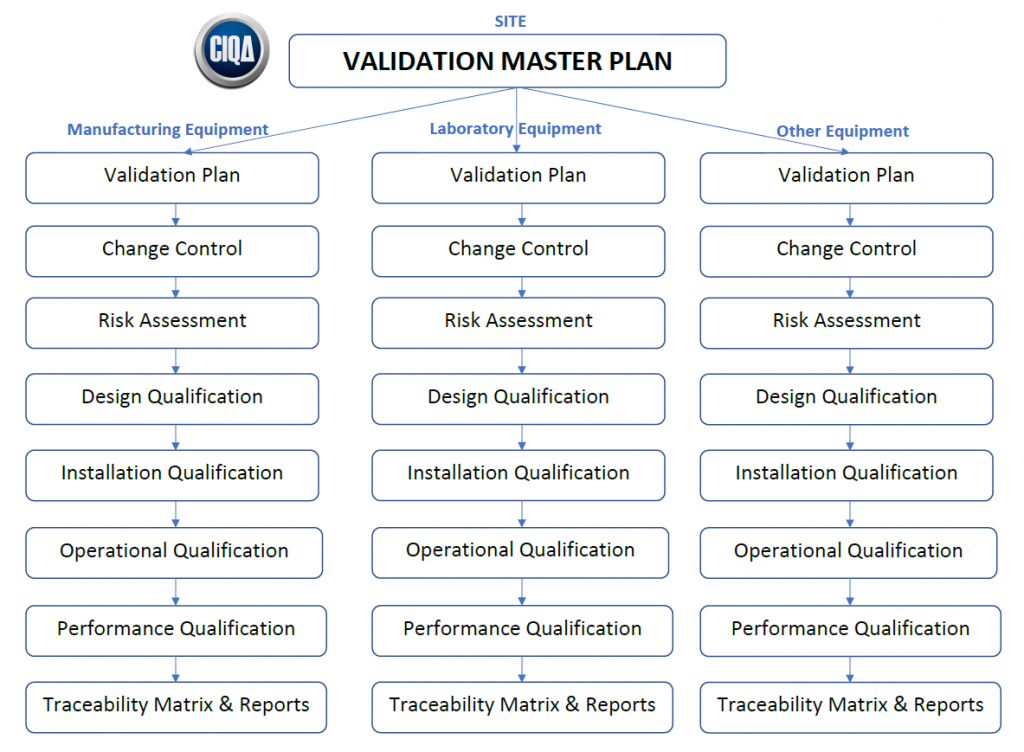
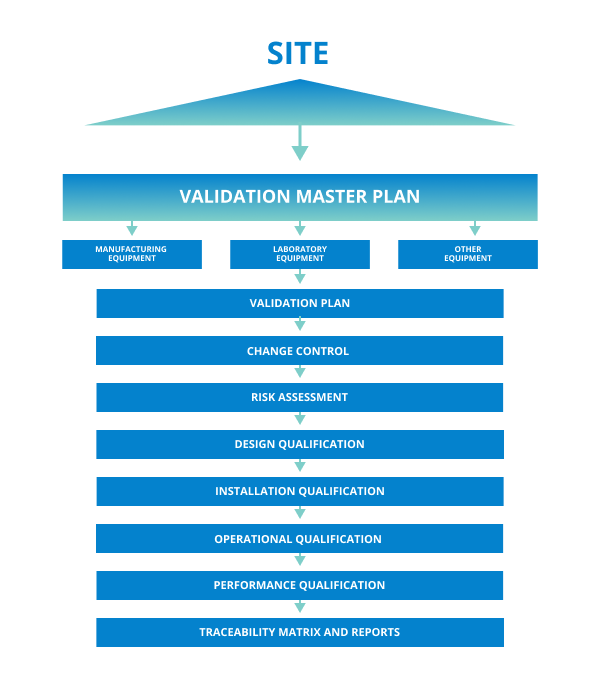
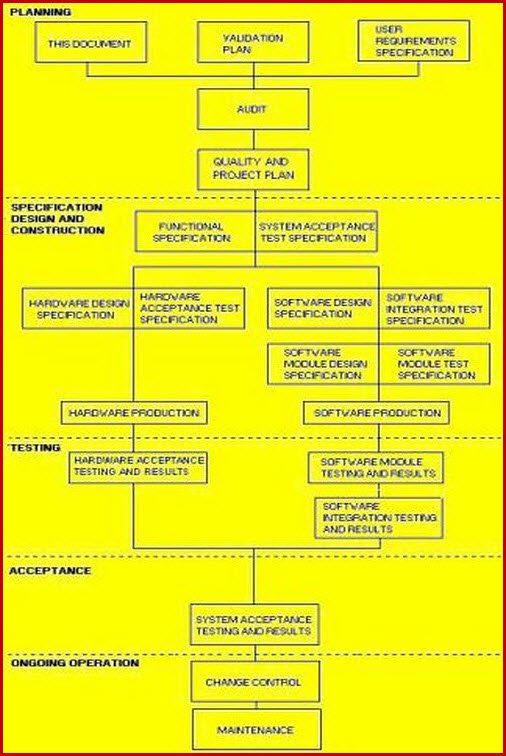
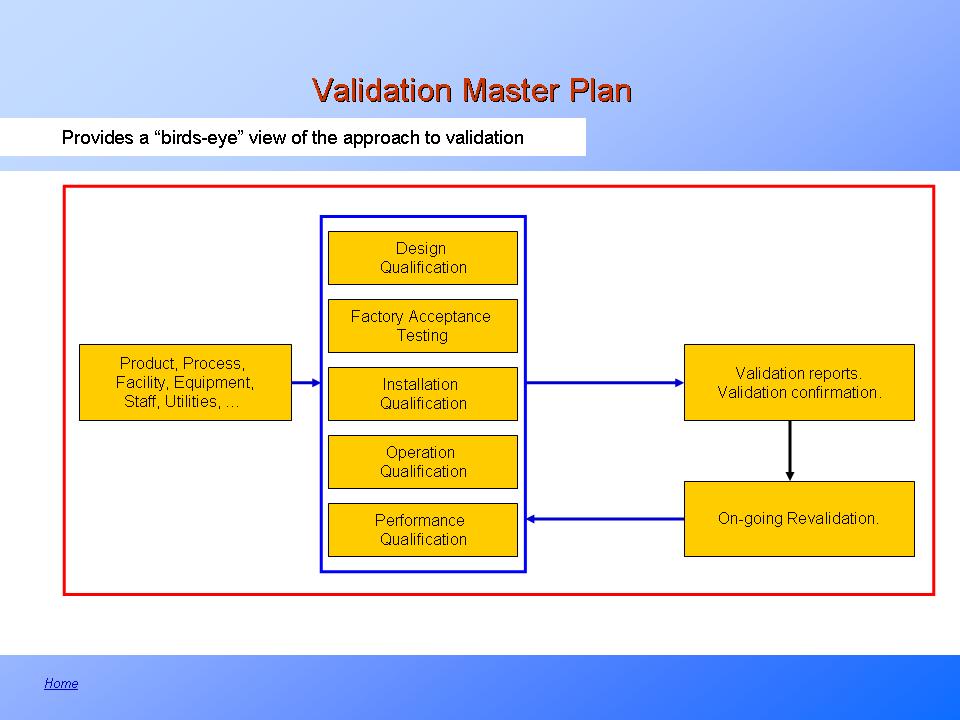
It defines responsibilities, prerequisites, and approaches for validating cleaning of equipment, products, and facilities. The first step involves defining the scope of the vmp. Web the purpose of the validation master plan template (vmp) is to describe the organization’s overall strategy, approach, and responsibilities for validation of computer systems and software. Items indicated “*” are listed as essential in rules and guidance for pharmaceutical manufacturers and distributors 2007, annex 15, 4. It can include information such as the scope of work, timeline, resources needed, risk assessment criteria, quality.
Web three (3) options to create a validation master plan. Web a validation master plan (vmp) outlines the principles involved in the qualification of a facility, defining the areas and systems to be validated, and provides a written program for achieving and maintaining a qualified facility. The purpose of this guideline is to provide guidance on the preparation of validation master plans (vmp). Web the master validation plan is designed to provide a planned and systematic framework within which all validation activities will occur. The purpose of this document is to record the schedule for conducting the validations and record the status and accomplishment of the validation plan.
Vmp is basically a summary of the validation strategy. Validation evaluates the software to ensure compliance with requirements. Web the purpose of this validation master plan (hereinafter known as the “plan” or “vmp”) is to provide guidelines and protocol for the validation of applicable processes, equipment, and software used in the production and verification of products manufactured by engineered medical systems, llc. Web the following template is suggested for a validation master plan which can be adapted for local use. Web the validation master plan is a summary of validation strategy.
Web fda quality systems regulations. It defines responsibilities, prerequisites, and approaches for validating cleaning of equipment, products, and facilities. The purpose of this document is to record the schedule for conducting the validations and record the status and accomplishment of the validation plan. Web use this template to: This includes identifying what needs to be validated, the areas of business impacted, and the necessary resources for validation tasks. Items indicated “*” are listed as essential in rules and guidance for pharmaceutical manufacturers and distributors 2007, annex 15, 4. The purpose of this guideline is to provide guidance on the preparation of validation master plans (vmp). Web the following template is suggested for a validation master plan which can be adapted for local use. All functions, departments and manufacturing sites within the sponsor or its contractors operating under gmp regulations or guidelines. Web the validation master plan is a summary of validation strategy. Vmp is basically a summary of the validation strategy. You can create a great protocol, using a template. In this comprehensive guide, we’ll address key questions such as “what does a validation master plan include?” and “how can i create a vmp?” and “who is responsible for preparing a vmp?” Web standard operating procedure & guideline for preparation and control of validation master plan (vmp) for instrument, equipment, facility in the pharmaceutical drug manufacturing plant. This document will also ensure that the manufacturing facilities comply with the local applicable gmp regulations and site requirements for validation.
Web A Validation Master Plan (Vmp) Outlines The Principles Involved In The Qualification Of A Facility, Defining The Areas And Systems To Be Validated, And Provides A Written Program For Achieving And Maintaining A Qualified Facility.
This document will also ensure that the manufacturing facilities comply with the local applicable gmp regulations and site requirements for validation. Web use this template to: You can create a great protocol, using a template. Web the purpose of the validation master plan template (vmp) is to describe the organization’s overall strategy, approach, and responsibilities for validation of computer systems and software.
The Validation Master Plan Also Includes An Overview Of The Processes That Support Validated Systems And An Appendix To Detail The Organization’s.
You can download a free sample of a validation master plan template in.pdf format. The purpose of this guideline is to provide guidance on the preparation of validation master plans (vmp). Web writing a validation master plan (vmp) requires a strategic and systematic approach. Web a validation master plan (vmp) is a document that outlines the overall validation strategy for an entire project including the systems and processes to be validated, the schedule to be followed, and the resources needed.
Validation Evaluates The Software To Ensure Compliance With Requirements.
Web a validation master plan (vmp) is a documented plan that outlines the overall strategy and approach for validation activities within a pharmaceutical or medical device manufacturing facility. To see the complete list of the most popular validation templates, click here. Web at the core of the validation process is a fundamental document known as a validation master plan (vmp). It defines responsibilities, prerequisites, and approaches for validating cleaning of equipment, products, and facilities.
Web The Master Validation Plan Is Designed To Provide A Planned And Systematic Framework Within Which All Validation Activities Will Occur.
Web three (3) options to create a validation master plan. Items indicated “*” are listed as essential in rules and guidance for pharmaceutical manufacturers and distributors 2007, annex 15, 4. Web standard operating procedure & guideline for preparation and control of validation master plan (vmp) for instrument, equipment, facility in the pharmaceutical drug manufacturing plant. In this comprehensive guide, we’ll address key questions such as “what does a validation master plan include?” and “how can i create a vmp?” and “who is responsible for preparing a vmp?”