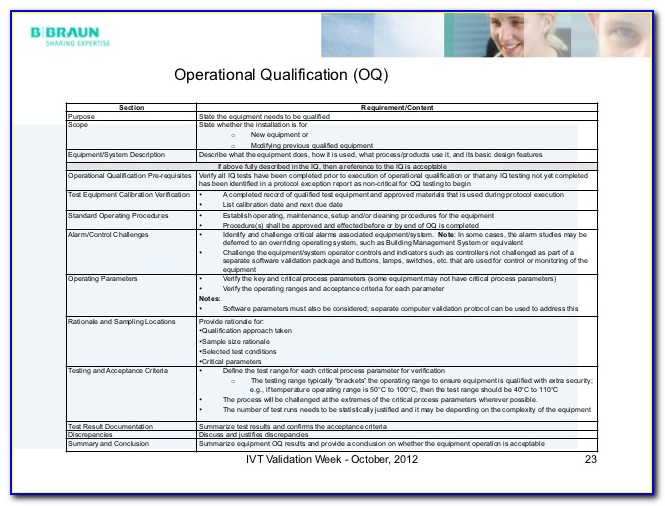
Web iq, oq, and pq stages are distinct steps in the equipment validation process. The key differences are in their specific objectives within that process. Web the objective of this protocol is to define the installation qualification (iq) and operational qualification (oq) requirements and acceptance criteria for the [insert system name and plant number] which will be located in the [insert area, packaging or manufacturing] at site [insert site name]. Tests the equipment to ensure it operates as intended within established parameters. Web iq, oq, pq protocols are methods for demonstrating that equipment being used or installed will offer a high degree of quality assurance such that production processes will consistently manufacture products that meet quality requirements.
The goal of process validation is to produce a stable medical device manufacturing process that offers consistent performance. Tests the equipment to ensure it operates as intended within established parameters. Web iq, oq, pq protocols are methods for demonstrating that equipment being used or installed will offer a high degree of quality assurance such that production processes will consistently manufacture products that meet quality requirements. These are the abbreviations we use in the medical device industry for the three steps of process validation: Web the objective of this protocol is to define the installation qualification (iq) and operational qualification (oq) requirements and acceptance criteria for the [insert system name and plant number] which will be located in the [insert area, packaging or manufacturing] at site [insert site name].
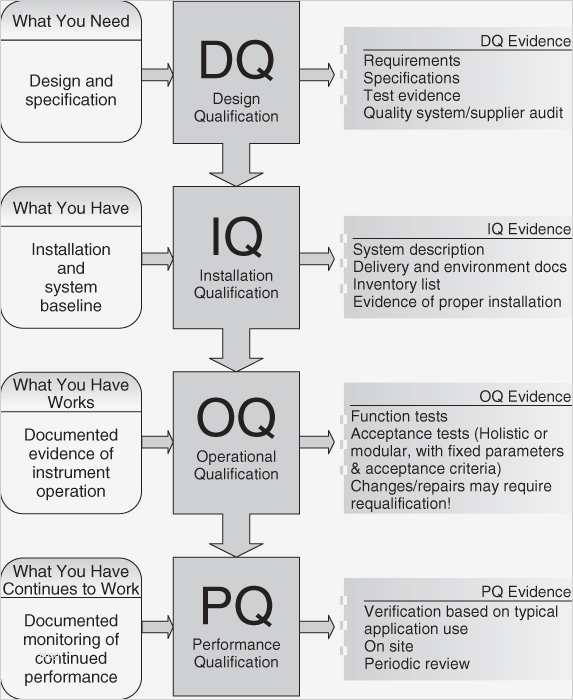
Installation qualification (iq), operational qualification (oq), and performance qualification (pq). Web understanding iq, oq, and pq for medical device manufacturing processes. Web write the objective of the protocol defining the installation qualification (iq) and operational qualification (oq) requirements and acceptance criteria for the equipment with location i.e., packaging or manufacturing, and the facility. Web what is iq, oq, pq? Tests the equipment to ensure it operates as intended within established parameters.
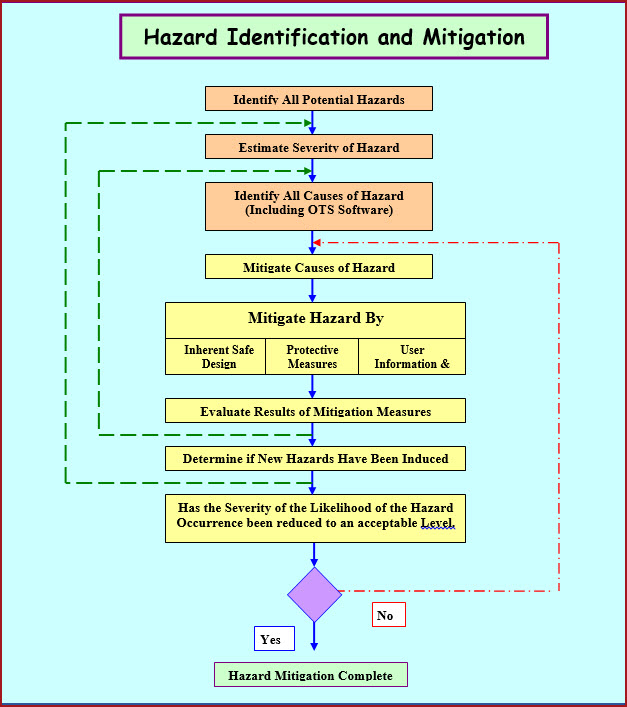
Web understanding iq, oq, and pq for medical device manufacturing processes. The goal of process validation is to produce a stable medical device manufacturing process that offers consistent performance. Tests the equipment to ensure it operates as intended within established parameters. Installation qualification (iq), operational qualification (oq), and performance qualification (pq). Each ensures the equipment’s dependability, functioning, and adherence to legal requirements in its own way. Web write the objective of the protocol defining the installation qualification (iq) and operational qualification (oq) requirements and acceptance criteria for the equipment with location i.e., packaging or manufacturing, and the facility. Web the objective of this protocol is to define the installation qualification (iq) and operational qualification (oq) requirements and acceptance criteria for the [insert system name and plant number] which will be located in the [insert area, packaging or manufacturing] at site [insert site name]. Use them right now to help with your qualification and validation projects. Web performance qualification (pq) demonstrate the process will consistently produce acceptable product under normal operating conditions. These are the abbreviations we use in the medical device industry for the three steps of process validation: Web iq, oq, and pq stages are distinct steps in the equipment validation process. Web what is iq, oq, pq? Web what is iq, oq, pq? Installation qualification (iq) for hardware verifies that the physical equipment and ancillary systems are installed correctly and in accordance with manufacturer specifications and regulatory requirements. Things to consider… • approved procedures and limits.
Things To Consider… • Approved Procedures And Limits.
Installation qualification (iq), operational qualification (oq), and performance qualification (pq). Web iq, oq, pq protocols are methods for demonstrating that equipment being used or installed will offer a high degree of quality assurance such that production processes will consistently manufacture products that meet quality requirements. Web iq, oq, and pq stages are distinct steps in the equipment validation process. Web write the objective of the protocol defining the installation qualification (iq) and operational qualification (oq) requirements and acceptance criteria for the equipment with location i.e., packaging or manufacturing, and the facility.
Web What Is Iq, Oq, Pq?
Web what is iq, oq, pq? Web the objective of this protocol is to define the installation qualification (iq) and operational qualification (oq) requirements and acceptance criteria for the [insert system name and plant number] which will be located in the [insert area, packaging or manufacturing] at site [insert site name]. Web performance qualification (pq) demonstrate the process will consistently produce acceptable product under normal operating conditions. The key differences are in their specific objectives within that process.
Installation Qualification (Iq) For Hardware Verifies That The Physical Equipment And Ancillary Systems Are Installed Correctly And In Accordance With Manufacturer Specifications And Regulatory Requirements.
Each ensures the equipment’s dependability, functioning, and adherence to legal requirements in its own way. The goal of process validation is to produce a stable medical device manufacturing process that offers consistent performance. Use them right now to help with your qualification and validation projects. These are the abbreviations we use in the medical device industry for the three steps of process validation:
Web Understanding Iq, Oq, And Pq For Medical Device Manufacturing Processes.
Verifies that the equipment is installed correctly, according to manufacturer’s specifications and site requirements. Tests the equipment to ensure it operates as intended within established parameters.