6.1 what is clinical evaluation? • evaluating data in terms of its suitability for establishing the safety and performance of the device. Web clinical evaluation report (cer) for medical devices: Web the clinical evaluation report (cer) provides clinical data to support the clinical safety and efficacy of a device. This document has been endorsed by the medical device coordination group (mdcg) established by article.
A guide for manufacturers and notified bodies. • evaluating data in terms of its suitability for establishing the safety and performance of the device. Stages in the clinical evaluation process. This document has been endorsed by the medical device coordination group (mdcg) established by article. The template is arranged into numbered.
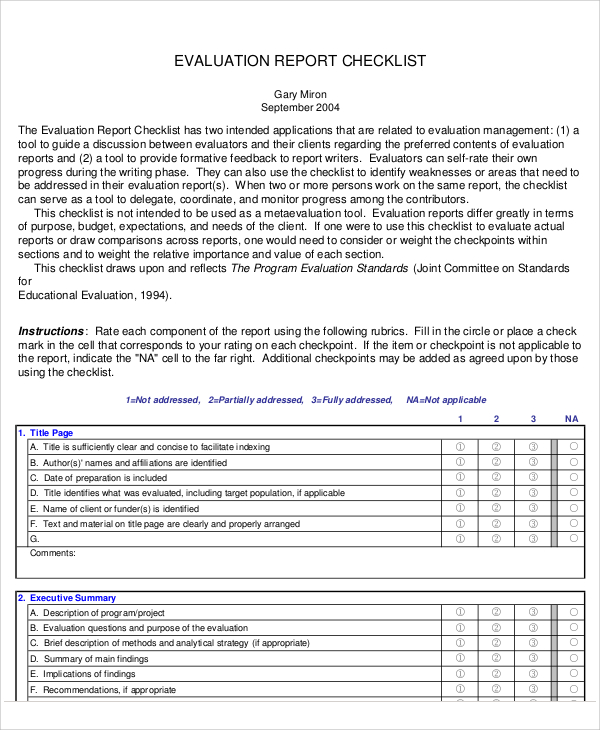
A guide for manufacturers and notified bodies. Web a clinical evaluation report (cer) documents the clinical evidence and conclusions for a medical device. • evaluating data in terms of its suitability for establishing the safety and performance of the device. This document has been endorsed by the medical device coordination group (mdcg) established by article. Web download the template for clinical evaluation assessment report, a document required by eu medical device regulation.
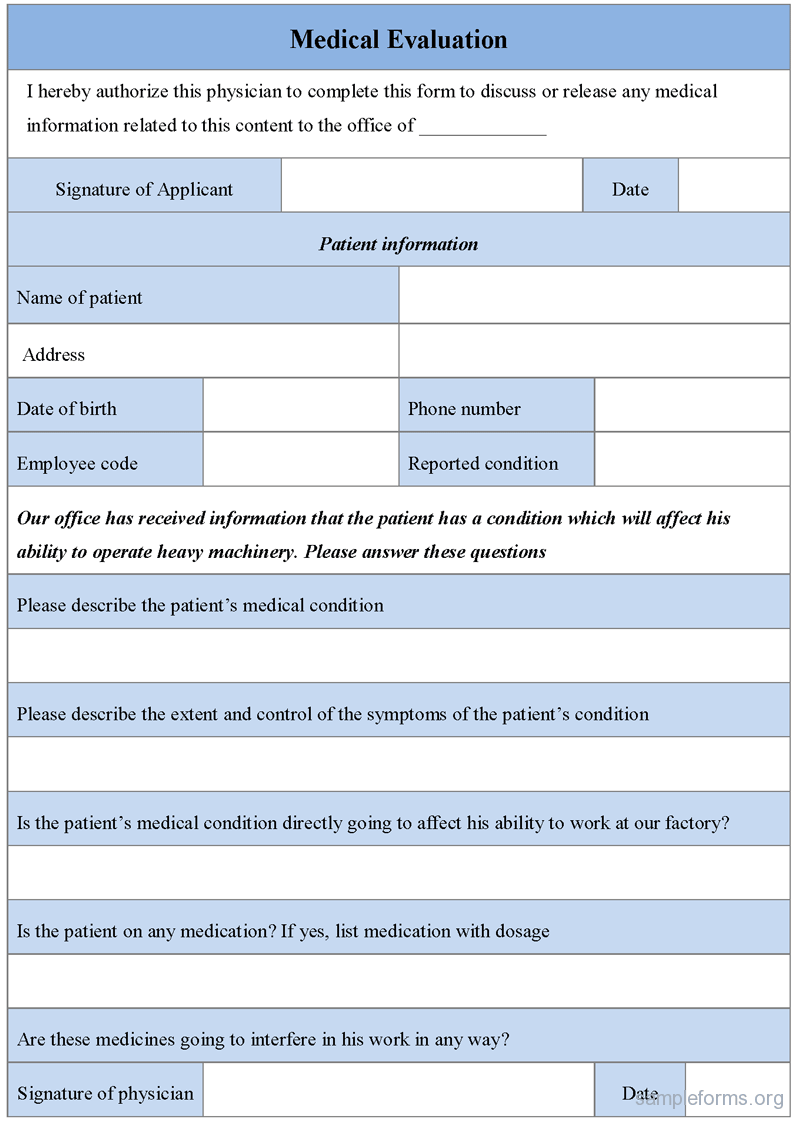
The initial clinical evaluation is the crucial step in effective mental health treatment. A guide for manufacturers and notified bodies. 6.1 what is clinical evaluation? Web the clinical evaluation report (cer) provides clinical data to support the clinical safety and efficacy of a device. General principles of clinical evaluation. Defining the scope and drafting a plan. The template is arranged into numbered. Web how to use this template. Web • identifying available clinical data relevant to the device and its intended use. Making sure cer evaluators are qualified. Web a clinical evaluation report (cer) is a comprehensive document that summarizes the results of the clinical evaluation process for a medical device. Web clinical evaluation assessment report template. Cancel anytime24/7 tech supportedit on any devicefree mobile app Web download the template for clinical evaluation assessment report, a document required by eu medical device regulation. Web check out our template walkthrough videos in which sebastian walks you through everything you need to know to perform your own clinical evaluation, post.
Learn The Steps, Sources, Elements And Updates Of A Cer For The Eu.
Web in this guide: Making sure cer evaluators are qualified. This page provides a range of documents to assist stakeholders in. Web clinical evaluation assessment report template.
Stages In The Clinical Evaluation Process.
It assesses the clinical risks associated with the use of a device, its. Web a clinical evaluation report (cer) provides clinical data to support a device’s clinical safety and efficacy. This document has been endorsed by the medical device coordination group (mdcg) established by article. It assesses the clinical risks associated with the.
The Template Is Arranged Into Numbered.
6.1 what is clinical evaluation? When certifying software as a medical device (or any medical device, really), you need a. Web download the template for clinical evaluation assessment report, a document required by eu medical device regulation. Defining the scope and drafting a plan.
Web A Clinical Evaluation Report (Cer) Provides Clinical Data To Support A Device’s Clinical Safety And Efficacy.
Web a clinical evaluation report (cer) is a comprehensive document that summarizes the results of the clinical evaluation process for a medical device. Web how to use this template. Based on the findings in the clinical data review as well as in the risk analysis, it can be inferred that the probability of a patient. Check out our template walkthrough videos in which sebastian walks you through everything you need to know.