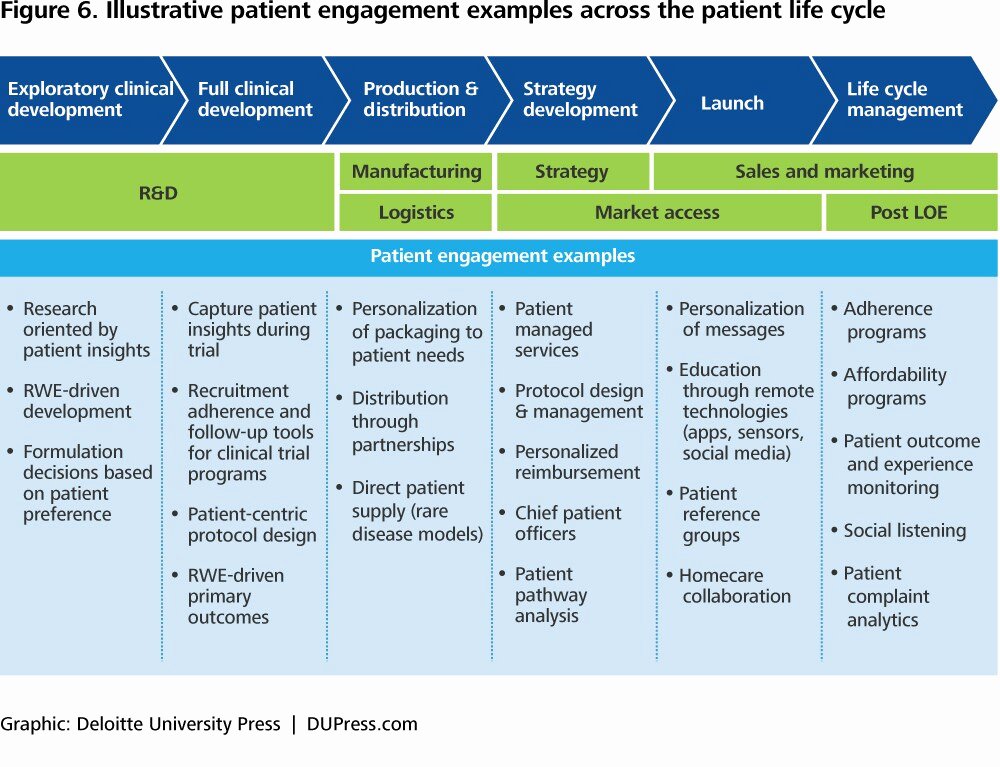
Tailor a clinical development plan to the specific needs of the treatment being developed. The clinical development plan (cdp) as per the medical device regulation (mdr) nhe. The concept should outline the specific objectives, study design, patient population, and endpoints of the clinical trial. The vision is transformed into distinct implementation phases and discrete steps, called clinical studies, each with well defined milestones and deliverables. What is a clinical development strategic plan?
What is a clinical development strategic plan? Explore precision's custom clinical development planning, tailored to novel study needs. The clinical development plan (cdp) is a new document that you must complete for the eu mdr technical documentation. A strategic, comprehensive clinical development plan (cdp) can help sponsors optimize efficiency, control costs, plan timelines, and maximize the probability of success for a new drug program. In this task, the clinical trial concept will be developed based on the identified target disease indication and literature review findings.
A strategic, comprehensive clinical development plan (cdp) can help sponsors optimize efficiency, control costs, plan timelines, and maximize the probability of success for a new drug program. In this task, the clinical trial concept will be developed based on the identified target disease indication and literature review findings. The clinical development plan (cdp) is a new document that you must complete for the eu mdr technical documentation. The vision is transformed into distinct implementation phases and discrete steps, called clinical studies, each with well defined milestones and deliverables. The concept should outline the specific objectives, study design, patient population, and endpoints of the clinical trial.
What is a clinical development strategic plan? Such a strategy encompasses various aspects of a program and can yield several benefits for a company, including: It should consider current knowledge about the condition being treated, the target patient population, and the regulatory landscape. The clinical development plan (cdp) as per the medical device regulation (mdr) nhe. The vision is transformed into distinct implementation phases and discrete steps, called clinical studies, each with well defined milestones and deliverables. In this task, the clinical trial concept will be developed based on the identified target disease indication and literature review findings. Advance global clinical development, with precision. This article is intended to assist medical device manufacturers and medical writers to leverage the cdp as a tool to showcase their clinical evaluation strategy and plan. The clinical development plan (cdp) is a new document that you must complete for the eu mdr technical documentation. Tailor a clinical development plan to the specific needs of the treatment being developed. The clinical development plan guide. A clinical development plan — a comprehensive strategy for developing an investigational product through regulatory submission — is a critical component of drug development and helps ensure that new therapies are safe, effective, and of high quality. A strategic, comprehensive clinical development plan (cdp) can help sponsors optimize efficiency, control costs, plan timelines, and maximize the probability of success for a new drug program. Explore precision's custom clinical development planning, tailored to novel study needs. The concept should outline the specific objectives, study design, patient population, and endpoints of the clinical trial.
The Clinical Development Plan Guide.
The clinical development plan (cdp) as per the medical device regulation (mdr) nhe. Advance global clinical development, with precision. Explore precision's custom clinical development planning, tailored to novel study needs. Tailor a clinical development plan to the specific needs of the treatment being developed.
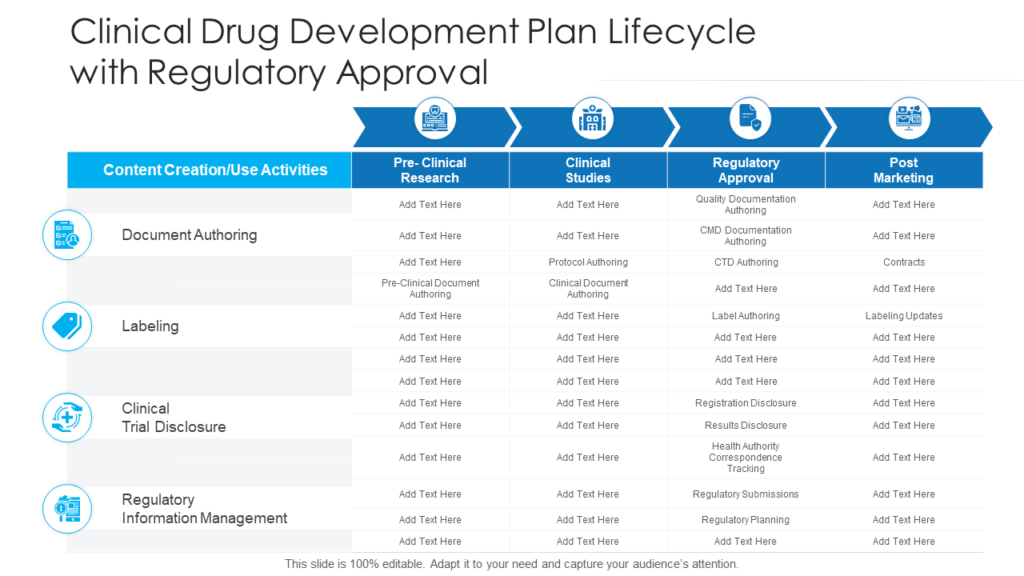
A Clinical Development Plan — A Comprehensive Strategy For Developing An Investigational Product Through Regulatory Submission — Is A Critical Component Of Drug Development And Helps Ensure That New Therapies Are Safe, Effective, And Of High Quality.
It should consider current knowledge about the condition being treated, the target patient population, and the regulatory landscape. Such a strategy encompasses various aspects of a program and can yield several benefits for a company, including: What is a clinical development strategic plan? The clinical development plan (cdp) is a new document that you must complete for the eu mdr technical documentation.
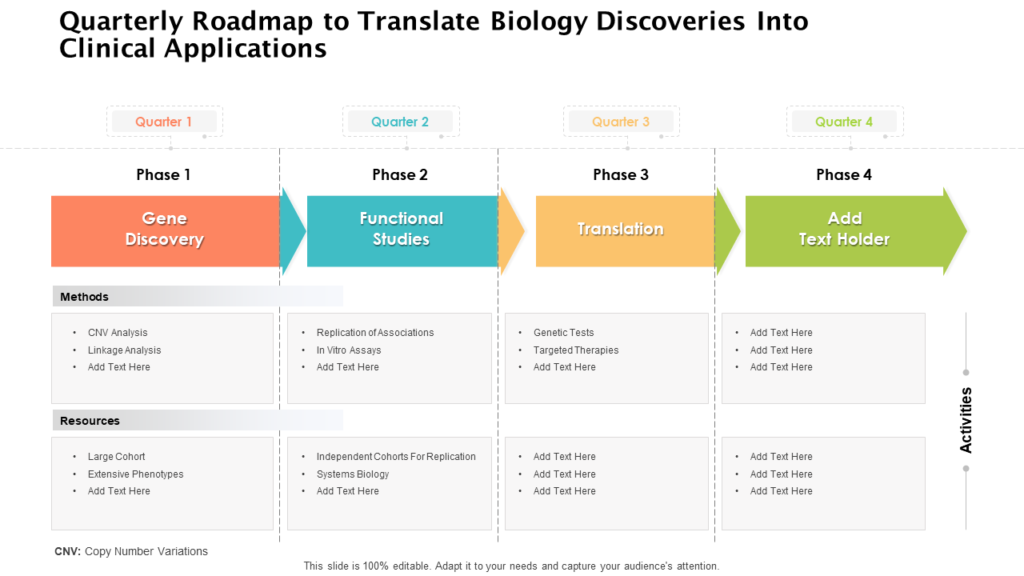
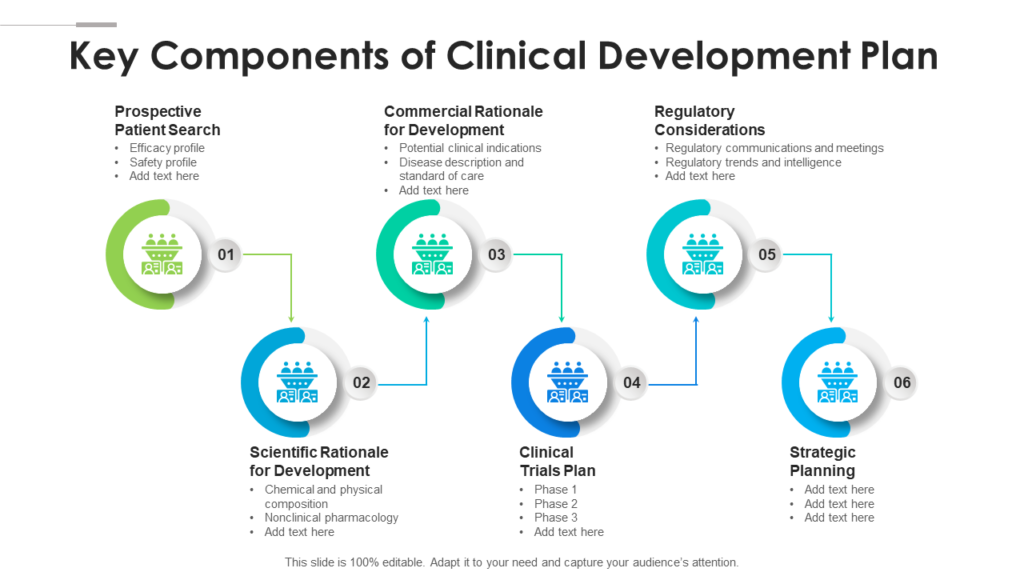
The Vision Is Transformed Into Distinct Implementation Phases And Discrete Steps, Called Clinical Studies, Each With Well Defined Milestones And Deliverables.
In this task, the clinical trial concept will be developed based on the identified target disease indication and literature review findings. There are two essential documents that any company developing a new pharmaceutical or biotechnology derived product requires to ensure it achieves its strategic goals. This article is intended to assist medical device manufacturers and medical writers to leverage the cdp as a tool to showcase their clinical evaluation strategy and plan. The concept should outline the specific objectives, study design, patient population, and endpoints of the clinical trial.